What is Ozone
The history of Ozone
The odor of Ozone was first reported by Van Mauren in 1785, in the vicinity of an electrical discharge. In 1840, Christian Schonbein observed a unique odor during electrolysis and electrical sparking experiments and identified this characteristic odor as a previously undetermined compound.
Ozone is named after the Greek word "Ozein", meaning to smell. However the identity of the structure of the compound was confirmed in 1867 as triatomic oxygen, simply mean the Ozone molecule contains three oxygen atoms, having the chemical symbol O3.
The Definition of Ozone
Ozone is the safest and second most powerful oxidant known to man. Ozone as an oxidant neutralizes contaminants or chemically alters them so that they can be more easily eliminated.
Chemistry of Ozone
Ozone (O3), a colorless gas, is the tri-atomic form of oxygen; also know as "Super Oxygen". It is a molecule consisting of three oxygen atoms (O1+O1+O1=O3).
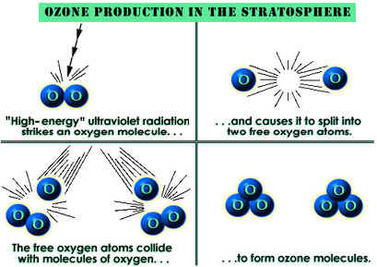
Ozone is formed naturally by the Ultra-Violet Rays of the sun (photochemical reaction) and by lightning (bio-electrical reaction). Ozone also formed synthetically by passing air or oxygen over a Ultra-Violet lamp (photochemical reaction). Some of the oxygen molecules split into two separate oxygen atoms. These single atoms then form semi-unstable bonds with the oxygen molecules (O2+O1=O3/Ozone). These oxygen molecules are highly reactive. This reactivity is because of the third atom of oxygen, also know as a "Hungry Atom". This atom being very eager to break away from this semi-unstable bond and react with any oxidizable compound (organic or inorganic). Also due to its reactivity, Ozone has a very short life span (about 20 min.).
The odor of Ozone was first reported by Van Mauren in 1785, in the vicinity of an electrical discharge. In 1840, Christian Schonbein observed a unique odor during electrolysis and electrical sparking experiments and identified this characteristic odor as a previously undetermined compound.
Ozone is named after the Greek word "Ozein", meaning to smell. However the identity of the structure of the compound was confirmed in 1867 as triatomic oxygen, simply mean the Ozone molecule contains three oxygen atoms, having the chemical symbol O3.
The Definition of Ozone
Ozone is the safest and second most powerful oxidant known to man. Ozone as an oxidant neutralizes contaminants or chemically alters them so that they can be more easily eliminated.
Chemistry of Ozone
Ozone (O3), a colorless gas, is the tri-atomic form of oxygen; also know as "Super Oxygen". It is a molecule consisting of three oxygen atoms (O1+O1+O1=O3).
Ozone is formed naturally by the Ultra-Violet Rays of the sun (photochemical reaction) and by lightning (bio-electrical reaction). Ozone also formed synthetically by passing air or oxygen over a Ultra-Violet lamp (photochemical reaction). Some of the oxygen molecules split into two separate oxygen atoms. These single atoms then form semi-unstable bonds with the oxygen molecules (O2+O1=O3/Ozone). These oxygen molecules are highly reactive. This reactivity is because of the third atom of oxygen, also know as a "Hungry Atom". This atom being very eager to break away from this semi-unstable bond and react with any oxidizable compound (organic or inorganic). Also due to its reactivity, Ozone has a very short life span (about 20 min.).
How Ozone was produce
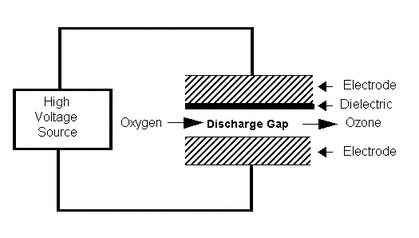
Using High Voltage Source, oxygen was convert into ozone which kills bacteria, mold, and viruses
The ozone will has a very short life span which will convert back into oxygen after half an hour
The ozone will has a very short life span which will convert back into oxygen after half an hour
Ozone (O3), is nature's strongest sanitizer and air purifier, it will:
- DESTROY: Cigarette, cigar, cooking, chemical, pet, and many other odors. Ozone can be used to neutralize smoke odors from fire damaged clothes and buildings, or a fireplace. Odors will be permanently neutralized leaving no residual chemicals or chemical odor.
- KILL: Bacteria and airborne viruses.
- REMOVE: Allergy causing pollen and microbials.
- PREVENT: Mold and mildew in Air Conditioning ducts, (sick building syndrome), the leading causes of Legionnaires Disease. There is little possibility that this type of odor will re-occur for a very long time.
- ELIMINATE: Toxic fumes from printing, plating processes, hair and nail salons, paints, and septic tanks. Many more.
Effectiveness
1. Eliminates harmful air-borne bacteria and pathogen in the air such as colliform, tuberculin and also the spore of the bacteria.
2. Eliminates foul smells such as cigarette smoke & toilet odor.
3. Prevention of "Sick Building Syndrome" (S.B.S) and improvement of alertness and work performance.
4. Improvement of indoor air quality thus helping respiratory of asthmatic patients, sinus and skin problems.
Applications:
Ozone Air Treatment Systems can destroy by oxidation the following airborne odors:
- Smoke
- Urine
- Mold
- Fungus
- Mildew
- Hydrocarbons
- Bacteria
- Hydrogen Sulfide
- VOC's
- Ammonia
- Algae
- Yeast
Ideal Usage
1. Commercial
- Office, Conference Room, Hotel, Entertainment outlet, Restaurant, Cinema & Theatre and enclosed areas.
- Library, School, University, Nursery, Kindergarden, Hospital, Clinic, Food Court, Public Toilet.
- Bedroom, Kitchen, Living and Dining Hall, Study Room, Toilet & Bathroom.
